“While the study did not demonstrate prasugrel was superior to clopidogrel in these patients, TRILOGY ACS provided some additional observations in this previously understudied population. Magnus Ohman, M.D., Duke Clinical Research Institute and Chairperson of the TRILOGY ACS trial. “TRILOGY ACS was designed to evaluate dual oral antiplatelet therapy in UA/NSTEMI patients who are managed medically without revascularization,” said E. 1 However, the rates of TIMI major or minor bleeding were higher in patients treated with prasugrel (3.3 percent of prasugrel patients versus 2.1 percent of clopidogrel patients HR=1.54 95% CI: 1.06-2.23 P=0.02). 1 In patients under age 75, non-CABG TIMI major bleeding occurred in 2.1 percent of prasugrel patients versus 1.5 percent of clopidogrel patients (HR=1.31, 95% CI: 0.81-2.11, P=0.27). 2 Results of this study were published in the New England Journal of Medicine and also presented during a late-breaking session at the ESC Congress 2012 (European Society of Cardiology) in Munich, Germany.įrom a safety perspective, TRILOGY ACS showed that rates of TIMI major bleeding events (including life-threatening or fatal bleeds) did not differ significantly between the prasugrel plus aspirin and clopidogrel plus aspirin treatment groups in patients less than 75 years of age or in the overall study population. Different from other large-scale trials, TRILOGY ACSPage (TaRgeted platelet Inhibition to cLarify the Optimal strateGy to medicallY manage Acute Coronary Syndromes) prospectively studied only the UA/NSTEMI population medically managed without revascularization (percutaneous coronary intervention (PCI) or coronary artery bypass graft (CABG) surgery). 1 This outcome was not statistically significant (P=0.21). 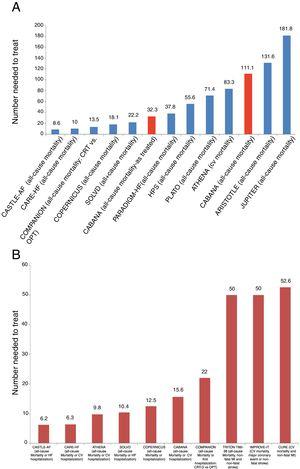
At 30 months, 13.9 percent of prasugrel patients versus 16.0 percent of clopidogrel patients experienced the combined primary endpoint of heart attack, stroke or cardiovascular (CV) death in patients under 75 years of age, the primary analysis population (HR=0.91 95% CI: 0.79-1.05). TIMI risk can be calculated on the TIMI website under "Clinical Calculators.Study did not meet primary objective of demonstrating prasugrel superiority over clopidogrel in this patient populationĭaiichi Sankyo Company, Limited, and Eli Lilly and Company recently announced data from the TRILOGY ACS study, a phase III trial comparing prasugrel plus aspirin to clopidogrel plus aspirin in patients with unstable angina (UA) or non-ST elevation myocardial infarction (NSTEMI), who were managed medically without an arteryopening procedure. 'TIMI risk' estimates mortality following acute coronary syndromes. 1 Pointĥ% risk at 14 days of: all-cause mortality, new or recurrent MI, or severe recurrent ischemia requiring urgent revascularization.Ĩ% risk at 14 days of: all-cause mortality, new or recurrent MI, or severe recurrent ischemia requiring urgent revascularization.ġ3% risk at 14 days of: all-cause mortality, new or recurrent MI, or severe recurrent ischemia requiring urgent revascularization.Ģ0% risk at 14 days of: all-cause mortality, new or recurrent MI, or severe recurrent ischemia requiring urgent revascularization.Ģ6% risk at 14 days of: all-cause mortality, new or recurrent MI, or severe recurrent ischemia requiring urgent revascularization.Ĥ1% risk at 14 days of: all-cause mortality, new or recurrent MI, or severe recurrent ischemia requiring urgent revascularization. % risk at 14 days of: all-cause mortality, new or recurrent MI, or severe recurrent ischemia requiring urgent revascularization. *Risk factors include: family history of CAD, hypertension, hypercholesterolemia, diabetes, or being a current smoker. Severe angina (≥ 2 episodes w/in 24 hrs).Known coronary artery disease (CAD) (stenosis ≥ 50%).TIMI Score Calculation (1 point for each): In patients with UA/NSTEMI, the TIMI risk score is a prognostication scheme that categorizes a patient's risk of death and ischemic events and provides a basis for therapeutic decision making. Among the group's most important works is the TIMI Risk Score, which assesses the risk of death and ischemic events in patients with unstable angina (UA) or non-ST elevation myocardial infarction (NSTEMI). Braunwald held the chairmanship until 2010, when he appointed Marc Sabatine to the position. The TIMI Study Group was founded by physician Eugene Braunwald in 1984. The group has its headquarters in Boston, Massachusetts. The Thrombolysis In Myocardial Infarction ( TIMI) Study Group, is an academic research organization (ARO) affiliated with Brigham and Women's Hospital and Harvard Medical School with a focus in the field of cardiovascular disease. For other uses, see Timi (disambiguation). For the IBM technology, see IBM i § TIMI.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
